![]()
Is your Company equipped to handle the challenges faced by many Bioequivalence organizations in today's global environment?
The challenges faced by Bioequivalence organizations across the world are many. Tight study deadlines measured in weeks (versus months), Silos of Information, Large volumes (Terabytes) of Data have led to high levels of Regulatory and Productivity stress. Adding to the challenge is the growing need for BE organizations to deliver hundreds of studies per year, up from only a handful of studies per year a few years ago. Current efforts vary from manual paper based systems to point based solutions that address part of the needs.
LIAS for Bioequivalence – An End to End solution
Megaware's Lab Informatics Automation System for Bioequivalence (“LIAS for BE”) is a web-enabled enterprise software platform for CROs and Pharmaceutical companies performing Bioequivalence studies. LIAS for BE addresses virtually all of the key requirements of a Bioequivalence organization; from Volunteer Registration, Protocol Management, Study Management, all the way to Documentation and Reporting. Wherever appropriate, the solution provides integration with best of breed third party applications, allowing customers to leverage their current investments and preferences.
LIAS – The Study Repository
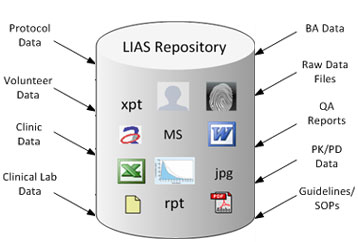 |
LIAS database acts a central repository storing all the Bioequivalence related information and study data. Using this LIAS repository, various departments can effectively share data between themselves. The repository ensures that reliable and consistent information is used by all departments and thereby bringing down the error potential while increasing process compliance
Highly Configurable
LIAS for BE, with its highly configurable approach has been designed to allow frequent changes to the deployed solution. Organizations can configure the LIAS solution to fit its processes and workflows. Changes can be made on the fly to the deployed solution without affecting the back end – thereby resulting in a minimal validation impact.
LIAS for BE Highlights
• Central Repository for all Bioequivalence and Study related information
• Highly Configurable solution that can fit the needs of organizations
• Compliance – Built to meet strict pharmaceutical and clinical regulatory requirements
• Enter Once, Use Many – Data once entered can be used in different parts of the Bioequivalence process; helping in
reducing transcription errors
• Comprehensive Audit Trail to track all the activities
• User, Group and Privilege Management to ensure hierarchical discipline
• State based checks to enforce the entry of mandatory information
• Report Generation and Print Control Policies for controlled circulation of reports
To know more about how our application can automate your lab process, please contact us
Contact Us
![]() +1.508.329.9375 (U.S)
+1.508.329.9375 (U.S)
![]() +91.22.66953120 (India)
+91.22.66953120 (India)
![]() info@megawareinc.com
info@megawareinc.com




