![]()
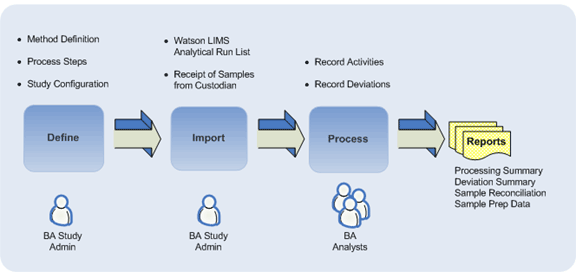
Sample Preparation is a critical component of the Bioanalytical workflow. LIAS BA Sample Preparation application tracks and manages samples through the sample preparation phase in a Bioanalytical laboratory. Users can Define the Sample Preparation methods and its process variables, Process samples while adhering to the defined method and Report sample data, process summary and deviations. This application is designed as an add-on to the industry standard Bioanalytical workflow system – Watson LIMS.
|
Samples from Watson LIMS Analytical Run List are imported into LIAS BA Sample Preparation application and follow an acknowledgement and review cycle before they can be used for further processing.
A study definition is created for each analytical run with appropriate sample preparation methods. These approved methods are then applied to the imported samples.
Activities related to the steps defined in the method are recorded such as - Process Start/End, parameter values and deviations, if any.
Configurable Approach
The system administrator can configure the application to map to your organization’s processes and terminologies. Once configured, the application can be used and reused until a change becomes necessary. Changes are easy and done in a compliant fashion. Studies, methods, execution modes (online/offline mode), log sheets, reports, privileges are examples of configurable items.
Compliance
LIAS BA Sample Preparation application is designed with regulatory compliance in mind. Compliance in a BA lab is greatly enhanced by deploying this application along with Watson LIMS. Data integrity and access is controlled through a granular set of task roles and changes tracked through Audit Trail. All this built upon industry standard Oracle RDBMS will help you comply with 21 CFR Part 11 compliance guidelines.
Key Benefits
• Addresses the need for sample tracking and reporting at the sample preparation phase
• Ensures adherence to a defined method
• Minimizes deviations and errors
• Generates relevant reports
• Enhances regulatory compliance in the BA Lab
• Integrates seamlessly with Watson LIMS
To know more about how our application can automate your lab process, please contact us
Contact Us
![]() +1.508.329.9375 (U.S)
+1.508.329.9375 (U.S)
![]() +91.22.66953120 (India)
+91.22.66953120 (India)
![]() info@megawareinc.com
info@megawareinc.com